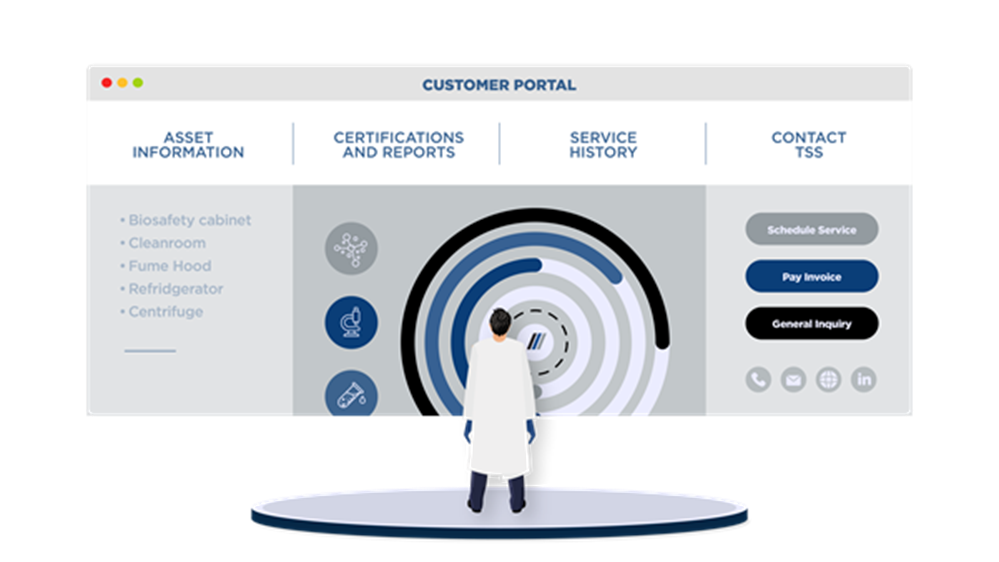
TSSConnect Data Access Solution
With a TSSConnect login and password, TSS clients can securely access, view and print their detailed service and certification records - 24 hours a day, every day. If you're not yet using TSSConnect, CLICK HERE to request access.
TSSConnect Benefits:
- Convenience: Everything you need in one place, available 24/7.
- Efficiency: Streamline your processes with quick access to key information.
- Support: Simplified communication with TSS experts.
TSSConnect Features:
- View and download reports
- Field Service Reports
- Lab Reports
- Formal Reports
- Access asset service information
- Service order details
- Asset information
- Service dates
- Contact TSS
- Schedule service
- Technical support
- Sales inquiries
Already signed up? Log into TSSConnect here.

