December 29, 2025
Outsourcing Calibration & Metrology Services
The technicians at analytical laboratories already have a lot on their minds. They work hard to ensure the testing they perform produces accurate and repeatable results. A major component of this is…
Read More
December 29, 2025
How to Calibrate a Centrifuge
A centrifuge is a vital part of a lab as it is used to separate components of a mixture based on density. Generally, this is essential in terms of research, diagnostics or production. For example, in…
Read More
November 22, 2025
Pass-Through Chambers: How Often to Clean and How They’re Certified
Pass-through chambers play a key role in contamination control by allowing materials to move between rooms without disrupting pressure relationships or exposing clean areas to unnecessary traffic.…
Read More
October 09, 2025
Finding the Right Calibration Solutions
Introduction to Calibration Solutions
The instruments and equipment that your company relies on for research and development, testing, and production are subject to wear and tear over the course of…
Read More
July 21, 2025
STERILIZER CHAMBER CLEANING PROCESSES
The Role of the Sterilizer Chamber in Laboratory Settings
To keep laboratory instruments clean, organizations typically use a sterilizer chamber, also known as an autoclave. They use steam to kill…
Read More
July 07, 2025
The Best Disinfectants for Cleanrooms and Controlled Environments
Cleanrooms and controlled environments require strict contamination control to protect products, processes, and patient safety. Disinfection is a core component, and it must be effective, validated,…
Read More
January 17, 2025
How To Develop A Validation Master Plan
Introduction to Validation Master Plans
In pharmaceutical manufacturing facilities, a validation master plan or VMP is a document companies use to ensure that they consistently create products that…
Read More
January 08, 2025
Mastering Equipment Qualification in the Pharmaceutical Industry
Introduction to Equipment Qualification in the Pharmaceutical Industry
Why is equipment qualification critical in the pharmaceutical industry? Your employees and customers depend on your…
Read More
January 06, 2025
The Critical Role of CQV in Pharma Quality and Compliance
Introduction to CQV in Pharma
What is CQV in pharma? Pharmaceutical companies implement commissioning, qualification, and validation (CQV) services to ensure product quality and patient safety.
Be…
Read More
September 16, 2024
Typical Autoclave Conditions for Sterilization
When you own or operate a pharmaceutical lab or cleanroom, your team needs to be aware of the typical conditions for autoclave sterilization, so you can adhere to industry best practices, following…
Read More
August 15, 2024
Cleanroom Cleaning Procedures
Why Proper Cleanroom Cleaning Is Crucial to Your Success
When you operate a cleanroom, it's imperative to maintain strict standards of cleanliness in the facility at all times. Whether you work in…
Read More
August 15, 2024
Understanding ISO Cleanroom Cleanliness Levels: Maintaining Standards in Your Cleanroom
How clean is the air inside your cleanroom? By following industry best practices and adhering to ISO cleanroom standards, you have a better chance of avoiding the disruption and delays that…
Read More
July 26, 2024
Vaporized Hydrogen Peroxide Decontamination: An Effective Method for Decontamination of Facilities and Equipment.
Decontamination is a cornerstone of infection control in healthcare facilities, laboratories, or other spaces that may contain biological hazards. Ensuring that equipment and surfaces are free of…
Read More
July 26, 2024
Understanding Chemical Indicators for Sterilization & Decontamination: How They Work and Their Importance
Sterilization is a critical process in healthcare, laboratory settings, and various industries to ensure the elimination of all forms of microbial life, including bacteria, viruses, fungi, and spores…
Read More
March 13, 2024
ISO Class 7 Cleanroom Requirements
What Is an ISO Class 7 Cleanroom?
An ISO Class 7 cleanroom is a controlled environment that adheres to specific cleanliness standards defined by the International Organization for Standardization (…
Read More
March 13, 2024
Air Change Rates And Maintaining Air Cleanliness
Cleanrooms or clean zones provide the necessary controls to reduce contaminants to a level appropriate for accomplishing contamination-sensitive processes.
Cleanrooms or clean zones can be…
Read More
December 04, 2023
How To Improve BSL-3 Laboratory Performance
When you own and operate a Biosafety Level 3 facility, it’s crucial that all facility systems and equipment perform their functions as you intend.
For example, it’s of utmost importance that the…
Read More
December 01, 2023
The MEP Commissioning Process for Biocontainment Laboratories
When preparing to work in Biosafety Level 3 (BSL-3) biocontainment facilities, your organization must adhere to biosafety guidelines, with a particular focus on mechanical, electrical, and plumbing (…
Read More
November 28, 2023
High-purity Water Testing – Where, When And How
Your company’s critical work to support cleanroom or laboratory applications depends on a reliable source of high-purity water. In pristine environments, such as pharmaceutical cleanrooms, you and…
Read More
November 28, 2023
Important Preventative Maintenance For High-purity Water Systems
In lab and cleanroom environments, high-purity water is essential for keeping operations going smoothly. The implication is that your organization must take seriously the importance of having a well-…
Read More
November 28, 2023
Types Of High-Purity Water In Labs
In laboratories and cleanroom settings, depending on the nature of the work you are doing, your operations will rely on different types of pure water. If you’re new to managing cleanroom…
Read More
November 01, 2023
What Is Computer System Validation In The Pharmaceutical Industry?
In the pharmaceutical industry, consistently ensuring the quality control of final products and compliance with regulatory requirements is of critical importance. One process that helps achieve this…
Read More
August 14, 2023
Understanding Lab Sterility Testing Methods
Sterility testing in lab settings helps to ensure that the proper conditions are in place for safe, accurate processes that meet industry standards and regulatory compliance requirements. These…
Read More
August 08, 2023
IMPORTANT LAB SAFETY EQUIPMENT
The critical work carried out in laboratory settings, especially labs for industries such as pharmaceutical, biotech, and other health- and medical-related fields, requires stringent safety processes…
Read More
August 04, 2023
Summary Of The USP <797> Updates
In November 2022, the United States Pharmacopeia (USP) announced updates to a number of chapters and guidelines, including USP <797>. After the 2022 announcement, these changes will become…
Read More
August 04, 2023
THE IMPORTANCE OF PARTICLE COUNTING IN LABS AND CLEANROOMS
Airborne particles in a controlled environment can have a significant negative impact on the integrity of the work performed and could even pose health risks. Airborne particle counting (AKA…
Read More
July 26, 2023
How To Approach Calibration Criteria
It is critical for equipment in laboratory settings to be properly calibrated in order to return reliable, repeatable results. Calibration helps to maintain accurate measurements, improve safety,…
Read More
July 11, 2023
High-Purity Water System Maintenance Guide
Water is used in nearly every aspect of laboratory processes, and it is critical that this water is of high purity — and in some cases, is essentially sterile — to produce safe, effective products…
Read More
June 13, 2023
SPECTROPHOTOMETER CALIBRATION AND MAINTENANCE
A spectrophotometer is one of the most common pieces of analytical equipment in a scientific laboratory. It measures the amount of light energy at specific portions of the spectrum as it passes…
Read More
June 13, 2023
Centrifuge Safety & Calibration
Centrifuges serve a critical purpose in laboratories, used to separate various components of samples for applications including purification, extraction of specific materials and concentration or…
Read More
June 06, 2023
Proper Biological Safety Cabinet Use
Biological safety cabinets (BSC) are critical equipment in laboratories to protect staff, research or samples, and the surrounding environment from contamination. These enclosed workspaces use…
Read More
June 05, 2023
Restricted Access Barrier Systems Explained
Pharmaceutical laboratories and manufacturing facilities have the ensure the cleanest, safest possible conditions to protect the public and their reputations. One of the most important elements found…
Read More
May 31, 2023
GUIDE TO LAB BALANCE CALIBRATION BASICS
Lab balance calibration is a critically important process in manufacturing settings, including pharmaceutical and medical products. Not only must daily calibration be performed to ensure accurate…
Read More
May 31, 2023
IQ OQ PQ FDA QUALIFICATIONS
The FDA takes qualification of lab equipment and validation of procedures very seriously in pharmaceutical and medical device research and manufacturing settings — and with good reason —failure to…
Read More
May 25, 2023
Quality Vendor Expectations
Too many of today’s vendors put profits ahead of service. By pushing multiple digital and automated self-service options, these vendors sacrifice service excellence, personalization and proactive…
Read More
May 24, 2023
Lab Freezer Validation Protocol
Freezer validation is an important process for pharmaceutical labs, medical manufacturing facilities and other life science and clinical markets where samples, drugs, raw materials and even vaccines…
Read More
May 11, 2023
BSL-3 Commissioning Requirements
Biosafety Level 3 (BSL-3) requirements apply to facilities that handle transmissible agents and toxins that can spread via the air and cause infection through respiratory inhalation. BSL-3 labs are…
Read More
October 31, 2022
Acceptable Cleanroom Materials
Keeping a cleanroom free from contamination requires attention to every last detail, including the materials that are allowed to be brought in and used in the cleanroom. Without a holistic, 360-…
Read More
October 12, 2022
Understanding HEPA Filter Pressure Trending And Why It Matters
High efficiency particulate air (HEPA) filters are an integral component of laboratory and cleanroom safety and integrity infrastructure. HEPA filter pressure measurements are one of the key factors…
Read More
October 12, 2022
The Importance Of Managing Cleanroom Air Exchanges Per Hour
Air exchange rate is a key term to understand in cleanroom construction, management, and maintenance. The air exchange rate for a cleanroom is defined as the number of changeovers of air within the…
Read More
September 28, 2022
Designing A Cleanroom Pressure Cascade
In cleanroom environments, control is critical, and air pressure is one of most important measures that must be closely monitored. Air pressure cascades are a highly effective way of controlling the…
Read More
June 27, 2022
How To Test Compressed Air Quality
Compressed air systems and compressed gas systems are one of the most critical utilities in a laboratory, food processing plant, cleanroom, or manufacturing environment. It can supply power,…
Read More
June 27, 2022
Preparing For Pharmacy Certification
Sterile compounding pharmacies encounter some of the most highly critical and sensitive challenges in keeping the work environment safe and compliant. There are two pharmacy certification types that…
Read More
June 27, 2022
Surface Sampling Methods For Microbiology
Environmental testing in controlled spaces is the most effective way to detect the presence of micro-organisms, which can present significant health and safety risks, especially in cases of…
Read More
June 15, 2022
Surface Wipe Sampling For Hazardous Drugs
In settings where sterile and non-sterile hazardous drugs and materials are regularly handled, constant vigilance is necessary in the ongoing effort to keep personnel safe while controlling…
Read More
June 03, 2022
Bag-In/Bag-Out vs. Non-BIBO Systems
High efficiency air filters are used in various industries and for many purposes. One such application is to use high efficiency filters to remove hazardous/harmful matter from exhaust air before…
Read More
May 18, 2022
Viable Environmental Monitoring And Trending Data
Environmental monitoring (EM) and testing in laboratory settings are key components in operating a lab successfully and safely. In order to remain safe — and compliant with regulations — facilities…
Read More
May 17, 2022
Which Is Better: Analog Or Digital Pressure Gauge?
Pressure monitoring devices represent a fundamental component of key media transport processes, including gas and liquid systems in refineries, electrical plants, industrial facilities, and more.…
Read More
April 26, 2022
What Does the CETA Organization Do?
The primary objective of the CETA organization is to promote quality assurance in controlled environment testing and certification. CETA also exists to disseminate knowledge, information and data,…
Read More
April 01, 2022
The Importance Of Autoclave Calibration And Validation
An autoclave is one of the most powerful pieces of equipment in a laboratory setting, drawing on pressure and heat to rapidly sterilize critical equipment more quickly and effectively than nearly any…
Read More
April 01, 2022
Understanding 'As Found' And 'As Left' Calibration Data And Its Importance
Accuracy and reliability are of great importance for lab equipment. For this reason, lab equipment must be calibrated on a regular basis — anywhere from monthly to yearly — in order to remain in spec…
Read More
March 22, 2022
Tips For Maintenance Of Laboratory Equipment
Most laboratories contain sophisticated equipment required to complete work with accurate, reliable, and repeatable results. Most of this equipment requires some form of maintenance or checks to keep…
Read More
March 17, 2022
Critical Aspects of TSS’ Certifications
Third-party certifications, whether company-wide or for individual employees, provide a differentiator, a competitive edge, and an assurance to which process and quality standards are consistently…
Read More
March 14, 2022
Biotechnology Cleanroom Considerations
Biotechnology is one of the most innovative areas in science today, focusing on all areas of life sciences: medicine, the environment, agriculture, the animal world, and more. These areas of…
Read More
February 16, 2022
How Often Should Test Equipment Be Calibrated?
The reality of using any type of equipment is that over time it will drift out of calibration. This is an understood and generally accepted part of owning and operating laboratory and other…
Read More
February 10, 2022
Cleanroom Protocols And Guidelines
Safety is paramount in the cleanroom, especially for those working in particularly hazardous environments and testing rooms. The best safeguards against unexpected incidents, emergencies and…
Read More
February 03, 2022
What Is Bioburden And Why Is It Critical To Assess It?
In the medical device manufacturing industry, proper sterilization is among the most critical steps in better ensuring the safety of the product for its end users. Since these devices are often in…
Read More
January 23, 2022
Use Of An Unidirectional-Flow, Clean Air Device (a.k.a. Clean Beach)
Rooms, or secondary engineering controls, are designed to mitigate contamination issues with samples or products, but sometimes a part of the process requires an additional level of…
Read More
January 20, 2022
Cleanroom Gowning Guide
In cleanroom settings, it is necessary to follow the proper protocols to maintain the integrity of the cleanroom and prevent contaminants from entering the controlled environment. Proper cleanroom…
Read More
January 18, 2022
The Importance Of Gloved Fingertip Sampling
Gloved fingertip sampling is a key test assessing the aseptic technique of compounding personnel. The gloved fingertip test assesses the amount of microbial contamination present on workers’ gloved…
Read More
October 25, 2021
How To Start A Cleanroom
Many professionals involved in the research and development of cutting-edge technologies are familiar with the importance of cleanrooms. Whether you use yours for R&D, manufacturing or some other…
Read More
October 23, 2021
How To Properly Set Up A Testing Lab Facility
Setting up a testing laboratory is a significant project, but it doesn’t have to be a daunting one. In this post, we’ll discuss types of test labs as well as the equipment and logistics that are part…
Read More
October 19, 2021
Commissioning A Lab: Why And How It's Done
The processes of building, opening or renovating a laboratory facility are unlike similar processes for just about any other application. While other kinds of facilities are typically planned,…
Read More
October 13, 2021
GMP Documentation Guidelines
What is GMP documentation? As a component of Good Manufacturing Practices, GMP documentation ensures the quality and traceability of manufacturing processes, maintenance and quality checks. The…
Read More
October 01, 2021
How To Avoid Contamination In A Lab
In a laboratory setting, contamination can wreak havoc in nearly every aspect of the operation — resulting in shutdown, cleaning, testing time, risk to personnel, skewed lab results, unusable…
Read More
September 02, 2021
How Often Should Biological Safety Cabinets Be Certified?
Biological safety cabinets are one of the primary safety measures in any laboratory setting that deal with microbes and infections agents. These secure, ventilated enclosures ensure that when…
Read More
September 01, 2021
The “Just Right” Guide to PEC Pressure Gauges
The pressure gauges installed onto primary engineering controls (PEC, such as biosafety cabinets or clean benches) aid in the assessment of proper function and troubleshooting when utilized properly…
Read More
August 24, 2021
Guide To Cleanroom Classifications: Understanding The Basics
Cleanrooms are used for the manufacture, testing and design of products in health- and safety-critical fields such as medical device manufacturing, pharmaceuticals, food and beverage, aerospace,…
Read More
July 06, 2021
Fume Hood Testing Procedure: Outlining The Basics
Fume hoods are designed to protect people who work with chemicals that may produce hazardous gases, vapors and particulates. They achieve this with airflow that pulls dust and fumes away from where…
Read More
June 14, 2021
8 Tips for Hazardous Chemical Handling In A Lab
Laboratory work with chemicals is a necessary component of a wide range of fields and industries: the medical field, pharmaceuticals, cosmetics, industrial applications, consumer products and many…
Read More
February 23, 2021
Why Endotoxin Testing Is Critical
Companies in the pharmaceutical and food industries must operate with extreme care to prevent their products from causing more harm than good. One of the most insidious threats these businesses must…
Read More
February 18, 2021
What Is Water For Injection?
Life as we know it on Earth would not exist without water. Not only is it one of the critical building blocks that make up the cellular structure of all living things, but it also serves virtually…
Read More
December 15, 2020
What Is Media Fill Testing?
Media fill testing is the method of assessing the technique and competency of personnel who manipulate and create compounded sterile preparations. To maintain the purity of samples, compounds and…
Read More
December 06, 2020
Considerations for Annual Cleanroom Shutdowns
Cleanroom shutdown events occur for numerous reasons, planned and unplanned. In returning to normal operation, there are key best practices that can be followed for a successful return to normal…
Read More
September 09, 2020
What Is ASHRAE 110?
Chemical fume hoods are an integral part of laboratory safety, and a malfunctioning hood could prove disastrous to workers interacting with dangerous chemicals and fumes. A trade organization called…
Read More
July 10, 2020
Understanding Purity And The Types Of Water Used In A Laboratory
No matter what type of work is done in a laboratory, it’s almost certain that lab water is required to carry out certain processes. Due to the sensitive nature of laboratory work — and the need for…
Read More
June 03, 2020
What Is Autoclave Sterilization?
What is autoclave sterilization? The simplest explanation is that it is a highly effective method of sterilizing and disinfecting objects through the use of heat — in the form of steam — plus…
Read More
June 02, 2020
The Importance Of Testing Lab Safety
Professionals in industries such as medical device manufacturing, biotechnology, healthcare, pharmaceuticals, nanotechnology and more are conducting some of the most vital work in testing labs.…
Read More
April 26, 2020
Lab Decontamination Challenges
If you are planning to reopen a lab after the Covid-19 crisis, please contact us and we are happy to help.
Lab decontamination is an important responsibility that comes as part of lab occupancy.…
Read More
February 06, 2020
What To Consider When Relocating Your Laboratory
Moving is seldom a stress-free process. That’s especially true when attempting to relocate a laboratory. While moving the contents of your home may be challenging enough, relocating a laboratory —…
Read More
January 30, 2020
How To Close A Laboratory Facility
Running a safe, compliant laboratory can be challenging enough. But what if you need to quickly learn how to close a lab? Decommissioning a working laboratory is far from simple. Numerous factors…
Read More
January 20, 2020
TSS Decommissioning Services
TSS is a marketplace leader providing comprehensive decommissioning, decontamination, lab closure, and relocation services to mitigate liability and assure compliance. Our team of Project Managers,…
Read More
January 13, 2020
Large Scale Decontaminations
Large scale decontaminations may be required for many different reasons – for example, a planned lab shutdown may include a space decontamination to facilitate planned maintenance, or an unplanned…
Read More
December 31, 2019
Viable Environmental Monitoring
Viable environmental monitoring provides critical information towards understanding the relative cleanliness of the sampled environment. Typically, viable airborne and surface data is collected to…
Read More
December 11, 2019
Quality for Service Providers
How would you define a high-quality testing service?
Quality means conformance to requirements; and the requirements are dependent on the related industry. This makes quality a subjective concept as…
Read More
November 13, 2019
HEPA FILTER INTEGRITY TESTING – CONSIDERATIONS IN HEPA FILTER PORTS
HEPA filters are sold in a variety of models and configurations, including with or without injection and testing ports. As HEPA filter leak testing involves upstream challenge of the HEPA filter with…
Read More
October 04, 2019
USP <795>, <797>, and <825> Postponed
USP has postponed the official dates of the below chapters from December 1, 2019 until further notice:
· <795>: Pharmaceutical Compounding – Nonsterile Preparations
· <797>:…
Read More
September 30, 2019
CDA (ISO 8573) - Why Test?
Why test CDA or CGS systems?
Compressed dry air or compressed gas systems are critical utilities in your clean environments. CDA or CGS systems may be used in process applications directly or…
Read More
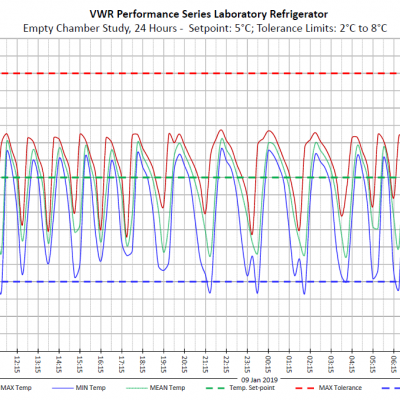
September 06, 2019
Validation: Temperature Controlled Units
Validation of temperature-controlled chambers is a critical component of a robust process and/or facility qualification plan. Validation includes Installation Qualification (IQ), Operational…
Read More
August 20, 2019
2019 Updates to USP <797> and how TSS is responding
USP <797> lays the foundation of standards for compounding sterile preparations. According to USP, “…<797> helps to ensure patients receive quality preparations that are free from…
Read More
August 06, 2019
Welcome to our blog!
Here at Technical Safety Services we are dedicated to the highest quality of compliance and safety for our clients, their processes and the people they serve. Our services are used by over 11,…
Read More